 Over the past year, our practice has seen an increasing number of patients for implant removal consultation. These are patients who have had implants in place for either cosmetic or reconstructive reasons. Patients have presented with a myriad number of complaints which have included many neurologic concerns not limited to memory loss, brain fog, dizziness, vertigo, confusion as well as physical ailments such as joint pains and muscle aches, GERD symptoms, kidney pain, sinus infections, bladder, and urinary issues, unexplained skin conditions, and rashes, even gout. Many complain of becoming increasingly debilitated due to chronic fatigue and have even suffered symptoms of depression and suicidal tendencies. In our professional experience, these are concerns that are becoming all too common in the breast implant patient.
Over the past year, our practice has seen an increasing number of patients for implant removal consultation. These are patients who have had implants in place for either cosmetic or reconstructive reasons. Patients have presented with a myriad number of complaints which have included many neurologic concerns not limited to memory loss, brain fog, dizziness, vertigo, confusion as well as physical ailments such as joint pains and muscle aches, GERD symptoms, kidney pain, sinus infections, bladder, and urinary issues, unexplained skin conditions, and rashes, even gout. Many complain of becoming increasingly debilitated due to chronic fatigue and have even suffered symptoms of depression and suicidal tendencies. In our professional experience, these are concerns that are becoming all too common in the breast implant patient.
Because these patient-reported symptoms tend to be more qualitative rather than quantitative. These concerns are oftentimes not treated with the appropriate level of attention by the plastic surgery community at large and the patients have aimlessly and unsuccessfully sought relief. Some sort of answers from their primary care physicians, specialists, blood work, and test after test with seemingly no end or hope for answers or a proper diagnosis to alleviate their symptoms and suffering.
Breast Implant Illness-Related Videos
Breast Implant Autoimmune Disease
However, a recent study has given some much-needed scientific support to these complaints suffered by many women with breast implants. The article is written by authors from the MD Anderson Cancer Center (Coroneos et al., 2018, Annals of Surgery) and reviews the long-term follow-up (large post-approval studies) data from the two primary implant manufacturers, Mentor and Allergan. Data from 99,993 patients are reviewed in this study. The study provides insight into the more systemic complaints associated with silicone and saline implants.
Data from the study demonstrates that in comparison with normative data, patients with silicone implants have higher rates of autoimmune disorders such as Sjogren’s syndrome, scleroderma, and rheumatoid arthritis. In 1992 the FDA prohibited the use of silicone breast implants for concerns that included cancer, connective tissue disease, and autoimmune disease. The Institute of Medicine went on to conclude that there was no evidence to support an association between breast implants and these systemic diseases. As a result, the FDA approved the current generation of silicone breast implants in 2006 for the two primary manufacturers, Mentor and Allergan. Despite the LPAS data being publicly available, this current study is the first to thoroughly analyze and report on this database.
Breast Implant Removal after Augmentation and Mastectomy
There is a troubling association in this study regarding the correlation between silicone breast implants and autoimmune diseases. Interestingly, both breast implant manufacturers are consistent with this LPAS data in reporting that more patient-reported rheumatologic symptoms are experienced by patients with silicone implants.
But it doesn’t stop their studies have also been found to associate silicone with a higher rate of stillbirth and melanoma skin cancer when compared to normative data. At seven years, the rate for breast revision surgery is 11.7% and 25% for breast reconstruction revision. Although nothing is 100%, this data cannot be ignored or even minimized.
An Ongoing Research to Address the Issues
The study does suffer from limitations. Chief among these limitations is that patients cannot be evaluated at an individual level so it is difficult to account for potential confounding conditions that could affect the interpretation of the data. Furthermore, data were collected under different protocols by both Allergan and Mentor, so a direct comparison between each company’s data is difficult.
Nonetheless, this data is important for plastic surgeons and patients to understand. Although many breast implant patients do not suffer from connective tissue or autoimmune ailments, there are certainly a significant number of patients with these concerns and the numbers are growing.
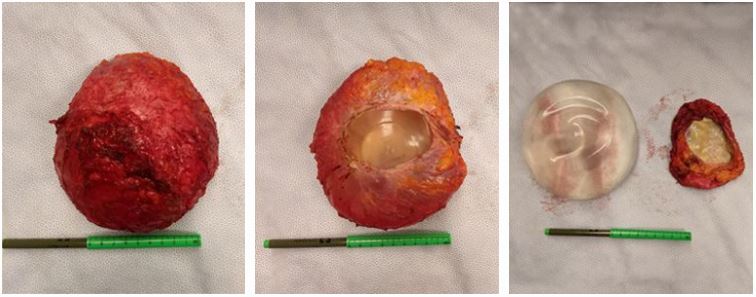
In our practice, our preferred method of treatment for women who come to us with complaints of unexplained suffering and the desire to ‘explant’ is an implant removal with en bloc, total capsulectomy.
Our protocol continues with sending the capsule for pathology, particularly if there is a concern for breast implant-associated ALCL lymphoma. It should be noted that in this particular study there has been one case of BIA-ALCL. On a lighter note, I have also found testimonials, too many to list, of increased energy, stamina, less joint and muscle pain, of the complete disappearance of unexplained suffering, and an undeniable amount of women insisting that the once unexplained decline in their health is on the mend. They have become the proof positive of something that undeniably needs further investigation.
So, as we continue to follow up on breast implant-associated conditions, we’re hoping to shed increasing light into what might increase the risk for some patients being more susceptible to developing these types of symptoms and what factors may make it safer for others. Either way, it is absolutely necessary to increase awareness of these conditions among patients and plastic surgeons so that together we can evaluate and hopefully alleviate the chronic suffering that too many patients have and are still too often experiencing.

30 thoughts on “Breast Implant Associated Autoimmune Disease Study Review”
WOW!! You are not my surgeon but I wanted to thank you . The FDA and many PS and PC are not being honest or are not informed. The validity and diagnosis of our illness is such a struggle for us ..this review is much appreciated.
Thank you for your input. I am glad that this information was helpful.
Thank you, I absolutely agree I am having kidney issues when I never had any before, fibromyalgia, hair loss, memory loss feet and hands swell different times of the day. I have many more symptoms. Thank you again Sir for your recognition of the damage silicone implants do I had a extracapsular and intracapsular implant rupture in 2019.
How can we get copies of these studies to support arguments for insurance coverage of explanation?
I have the Allergan silicone for 7 years now. I have some symptoms listed in the article and more. I want to be sure that these are from the implants before removing them. Is there a way to do this?
I would suggest seeing your primary care doctor and asking for a referral to a rheumatologist. A rheumatologist is a correct physician to order tests in order to determine the underlying cause of an autoimmune disease. Thank you for your question.
Dr Tiwari
About six months after my implants reconstruction from dounless mastectomy I came back to you and said I wanted the implants removed. I was and still have allergy issues digestive issues and had the sense the implants were stressing my body. At the time you convinced me that was not possible because the capsule forms around them and protects my body. You also said I would be very unhappy with the results because I would be concave vs flat after the surgery. I have continued to suffer from lethargy I used to be very active. But have also been under extreme stress from business for 5 years so it is easy to write off to that. But my body still is very sensitive and break out in hives at small things such as wrong laundry detergent that I did not have before and significat digestive issues. I recently saw this article from you and connected to a BII group on Facebook. Many of their symptoms seem like my own. In general if I hadn’t come to you just after the surgery with my intuition that something wasn’t right it would be easy to write off my issues tonthe extreme stress of my life and move on. But I am wondering if your feeling around the effects on the body have changed given this new research.
Thank you for reaching out to me. Over the past five years, my understanding of breast implant illness, which can also be characterized as ASIA (autoimmune/inflammatory syndrome induced by adjuvants), has increased greatly. This is largely based on two relatively recent publications in the scientific literature. The first is from MD Anderson Cancer Center, where I trained, and it categorizes the data from the long-term follow up studies that the FDA has mandated to the two primary implant manufacturers. The second is a paper published in 2017 from the Netherlands that looked at 30 years of implant data over 100 patients and confirmed findings from prior historical studies. I have this paper listed below. In short, my understanding and appreciation for this condition has most certainly evolved and changes over the past five years. I regret that when we last met, I did not have a better understanding of the scientific literature on this topic, nor was I equipped to fully understand the magnitude of your concerns. If you wish, I would be happy to see you once again and feel certain that I have more to offer in this regard than in the past. Please feel free to reach out to us at info@mwbreast.com to schedule a visit with me.
Immunol Res. 2017; 65(1): 120–128.
Two hundreds cases of ASIA syndrome following silicone implants: a comparative study of 30 years and a review of current literature
Maartje J. L. Colaris,#1,2 Mintsje de Boer,#1,2 Rene R. van der Hulst,1,2 and Jan Willem Cohen Tervaertcorresponding author1,3
Thank you I have scheduled to see you April 25
facebook type in search – breast implant illness healing warriors
21 years sick with textured saline implants but was all in my “head” So glad to see this finally coming out!! A year and 4 months out from explant and 85% better!
Can you tell me do you believe it is possible that Celiac Disease, an autoimmune disease could be a result of silicone breast implants related ASIA ?
I was diagnosed with Celiac a year after having my implants in but according to genetics I do not carry the gene/trait.
I am working on approval for removal.
Dr. Pankaj Tiwari,
I’m 56 years old and explanted April 13th, 2018, with 12 year old Mentor Spectrum Smooth Saline Adjustable Implants. Prior to Explant I had an array of illness including, Neurological, Pulmonary, Abdominal, bone, joint, and muscle issues. I also had weight loos greater than 40 lbs, premature aging, vision, hearing loss, brain fog, memory issues, BPPV, POTS, scattered granulomas throughout my lungs, liver, spleen,. I had multiple biopsies over the course of two years with my entire body covered with lesions tested positive for CD 3, CD20 positive Rare B-Cell lymphocytes. My onset of illness was February 26th, 2017. I wound up losing my job, insurance, and home to this illness.
My Explant surgeon 👩🏼⚕️ (from Hawaii) was supposed to have done more testing, biopsies, and removing any tissues left by capsules. However, she did not do all these.
I had one breast tissue sample, one capsule sample, no biopsy of lymph node. I also had a small preformation(hole) in right Implant. My lymph nodes have been swollen for as long as I’ve sick and still are. I’m still having breast pain due to lumps, cysts, in my breast. I continue to have lymph node swelling in ancillary, throat, neck, clavicles, and pelvic, and abdominal area. I’m seeing an ENT, GASTROINTESTINAL, NEUROLOGIST, And a Pulmonologist March 28th. I’ve just had Endoscopy and Colonoscopy with abnormal results. My gut issues got worse after Explant as did the inflammation. My fatigue and dizziness, pain, memory issues associated with my breast implants disabled me literally. Finally on July 3rd, 2018, I was found to be 💯 % disabled. I continue to struggle with breast implant illness, I myself have always believed that I have some type of Lymphoma associated with my implants. Now as another issue is Heavy Metal Toxicity from MRI CONTRAST DYE. Multiple granulomas started coming up, bone, muscle, hip, pelvic, vibrations, long bone pain arm/legs, my feet, biceps.
I just want help, complete testing, and removal of everything left behind from my Explant surgery. I still have my implants, tissue slides, but they threw my capsules away, because I had Smooth Implants no need to test for BIA ALCL, three months later they threw them away. I had previously ask about the capsules and they told me two to three years. I have pictures, reports, CD’s of all Images ULTRASOUND, CT, MRI’s.
I just need help.
Thank you 🙏 for you hard work and dedication to trying to help the other medical community understand the importance of BREAST IMPLANT ILLNESS AND BIA ALCL.
An interesting discussion is worth comment. I do believe that you ought to write more on this
topic, it might not be a taboo subject but typically people do not
discuss such issues. To the next! Best wishes!!
Thank you. We plan to publish more related articles and will be sure to share once it’s out.
I got my breast implants 18 months ago and I was diagnosed with arthritis 3 days ago.Do you think there is a connection between these events. How can I find out, because if that is the case I would get my implants removed
In your experience, does the symptoms of autoimmune disease (autoimmune enteropathy and autoimmune hepatitis) improve when implants are removed?
Breast cancer survivor here. In the last 4 years, went from hiking 700 km, traveling the world, rappelling down waterfalls in Costa Rica to sitting on the sofa. Despite exercising in the pool three times a week and lifting weights with a trainer twice a week, I am exhausted and can’t even walk up the stairs without extreme shortness of breath and all over muscle and joint pain. !4 year silicone implants coming out this year. As a physician’s wife, I especially applaud you for highlighting the dark side of this lucrative cosmetic business. Implants have no place in the human body. Thank you for your advocacy. By speaking out, you’ll change women’s lives and best practice in breast reconstruction.
What an informatative yet understandable writing! So many people dont seem to have the patience to get to know the ins and outs of this issue. It is very real and your dedication to educating the public is so appreciated!! There is so much more to all.of this that will be discovered for years to come. Thank you for your valuable information!
I had Meme, polyurethane covered implants for 26 years that had ruptured and replaced them with Allergen silicone smooth implants in 2016. I just had total Capsulectomy last week to remove the Allergen.
I have Sjogrens Syndrome diagnosed 23 years ago, thyroid and kidney issues. Thank you for the information. I am hopeful the recent explant will help resolve some health issues.
I suffered for eight years with unexplained muscle weakness and stiffness, to the point where I couldn’t walk, hold a cup, and I even had to sleep sitting up because of weak lung muscles, because I couldn’t breathe lying down. I had a silicone Lap-band removed and I am now back to normal. When the doctor removed it, he said he had trouble because it looked like it had melted, and was stuck to my stomach. Obviously my body was attacking it as a foreign body. I also had a reaction to a silicone cream. There needs to be a test for an allergy to silicone and other materials used in any implants.
Great information! I have been carrying a saline tissue expander (silicone shell) in my chest for 4 years because I didn’t trust any of the implants or my plastic surgeon. Because of the latest research – like what you have published here – I have decided to have the expander removed and “go flat” or leave a little fold of skin or whatever it takes to NOT look concave. Any suggestions? Thanks.
I had explant surgery 14 months ago. I still have brain fog but most symptoms have decreased. My Dr was unable to remove all of the capsule and said it can be dangerous to try to remove because how thin is was. Must the capsule be completely removed for a full recovery?
I’m having the same issues, few years after breast implants I developed rheumatory arthritis/auto immunes. I am considering an explant, however, I went from an A cup to a D cup and do not want to go back to being flat chested. Are there any other options?
10 year survivor suffering from severe nausea for the past 15 months. I have a ruptured left implant that no one seems worried about when I call to book an appointment. I have been offered appointments for summer 2020. I have had so many tests done and surgeries done. Thank you, Dr. Tiwari for caring and pursuing this issue about our implants. I’m hoping to get an appointment with you soon.
My first set of textured implants(Allergan textured) were placed in 1997, when i was 27 years old. 1999, I started to have autoimmune issues (Raynauds, GERD, antral varices, difficulty swallowing, and it was chalked up to “just unlucky genes” making no link to the implants. In 2001 I was diagnosed with CREST Syndrome. (diagnosed by Hal Scofield at the University of Oklahoma Medical Center). Problems sustained at a moderate level over the next 12 years, primarily severe gastrointestinal and severe Raynauds in hands, feet, tongue. Capsular contraction began, pain, hardness, and signifiant changes in breast shape. In 2016, they were removed (implant plus capsulectomy due to the infection, calcium chards, and contraction) and replaced with Sientra textured “Gummy Bear” implants. One year later, In 2017 I had a severe LSCs (Limited Cutaneous Systemic Sclerosis aka Limited Scleroderma) reaction/involvement. My heart, lungs, nerves in scalp, legs, and arms were inflamed and painful.. Swallowing, breathing, walking, were difficult. Joint pain and muscle weakness dominated my life. In 2019, just 6 months ago, contraction started again. My implants cause significant pain daily. I am seeing my surgeon to have them removed.
Omg!!!! I am so excited to FINALLY figure out what is wrong with me!!!!
I got saline implants May 2004 when I was 28. I still have them in.
I’m so tired of spending all of this money on doctors who just don’t understand.
What is my next step?
Thank you, Christy. Please contact our office at info@mwbreast.com and we can walk you through your options and next steps. Look forward to hearing from you!
Thank you, Dr Tiwari, for your candor on the subject of BII. In the summer of 1982 I had silicone implants for cosmetic reasons. Within 8 yrs I developed debilitating pain. Eventually diagnosed with FMS. I have seen many specialists and have been tested for a variety of autoimmune diseases as well as a host of other illnesses that may cause my symptoms. Nothing. Chemical sensitivities have nearly ruined my life. I have multiple joint damage from unexplained inflammation diagnosed as osteoarthritis. In 2017 an MRI revealed multiple signs of rupture in both implants. My left breast started to have twinges of pain in 2013. Now my entire left side, ribcage area and down my left arm hurt. Nerve pain keeps me awake all night. I am desperate for a surgeon with experience removing these enbloc. I have an appointment with you in April in hopes that you can help me. We are trying to get insurance to atleast cover any complications that may arise after explant. As of now they refuse. This is the main reason I have not had my implants removed. For 30 yrs my life has been a constant struggle managing pain. I am exhausted. Please, can you tell me if you have had any success getting insurance to cover cases like mine? Thank you!
Im now just discovering what might be wrong with me. I have had mine in for 31 years, Yep..31 years. It is just in the last 10 years that I have had breathing and fatigue issues. Have no energy to do anything. Test after test uncovers nothing. They assume it’s aging and allergies. Anyway, I went from a AA size to a full B/small C. Aesthetically, do you have options for my situation. Im in Ohio.